Vapor pressures of H 2 O and HCl over the magnesium chloride hydrates... | Download Scientific Diagram
Minerals | Free Full-Text | Calcium Chloride-Assisted Segregation Reduction of Chromite: Influence of Reductant Type and the Mechanism | HTML
![Vapor-Pressure Measurement of Ternary System CaCl2 + [Emim]Br + H2O, CaCl2 + [Emim]Cl + H2O, CaCl2 + [Emim]Ac + H2O, and CaCl2 + [Emim]NO3 + H2O | Journal of Chemical & Engineering Data Vapor-Pressure Measurement of Ternary System CaCl2 + [Emim]Br + H2O, CaCl2 + [Emim]Cl + H2O, CaCl2 + [Emim]Ac + H2O, and CaCl2 + [Emim]NO3 + H2O | Journal of Chemical & Engineering Data](https://pubs.acs.org/cms/10.1021/acs.jced.0c00829/asset/images/medium/je0c00829_0004.gif)
Vapor-Pressure Measurement of Ternary System CaCl2 + [Emim]Br + H2O, CaCl2 + [Emim]Cl + H2O, CaCl2 + [Emim]Ac + H2O, and CaCl2 + [Emim]NO3 + H2O | Journal of Chemical & Engineering Data

Measurements of Vapor Pressures of Aqueous Solutions in the NaCl–KCl–H2O System from 493.15 to 693.25 K in a Fused Silica Capillary High-Pressure Optical Cell | Journal of Chemical & Engineering Data

ファイル:Properties of aqueous mixtures of pure salts. Thermodynamics of the ternary system- Water-calcium chloride-magnesium chloride at 25 ?C (IA jresv70An4p305).pdf - Wikipedia

SOLVED:Which of the following aqueous solutions has (a) the higher boiling point, (b) the higher freezing point, and (c) the lower vapor pressure: 0.35 m CaCl2 or 0.90 m urea? Explain. Assume

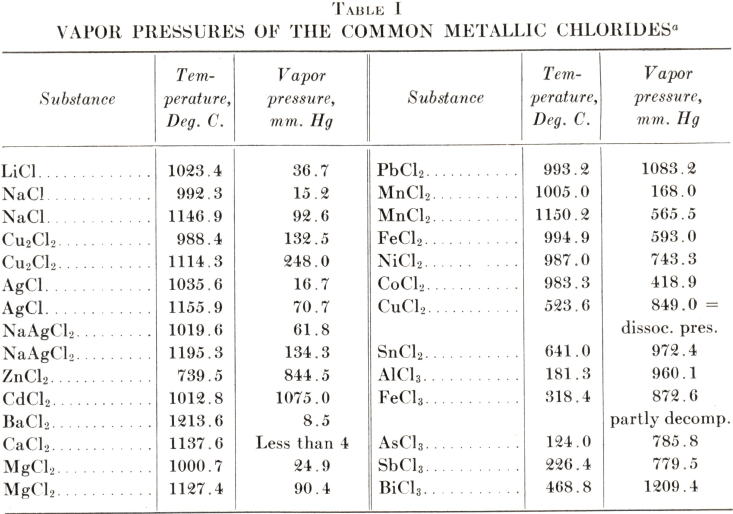
PDF) Thermodynamic consistency of solubility and vapor pressure of a binary saturated salt + water system. II. CaCl2 + H2O
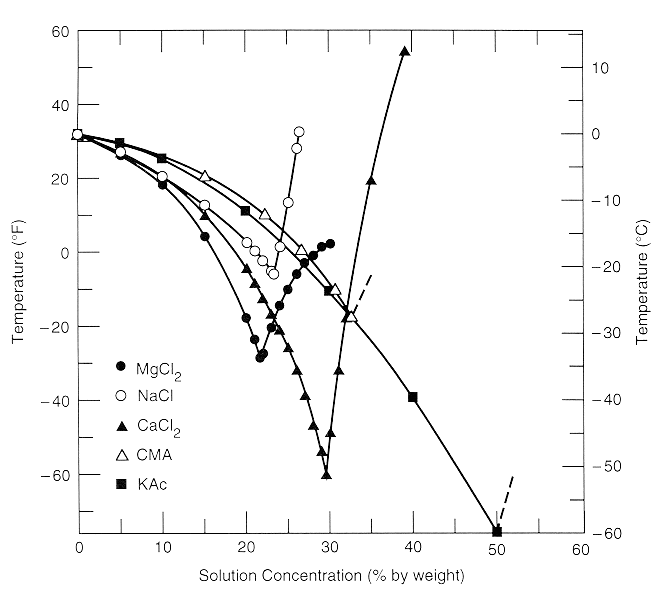
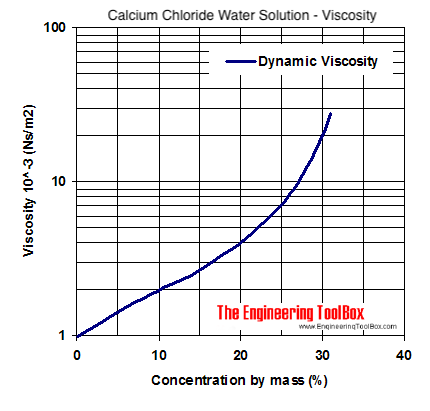
Recommended Anti-Icing Practices - Manual of Practice for An Effective Anti-Icing Program , June 1996 - FHWA-RD-95-202

Equilibrium vapor pressure over aqueous solutions of NaCl (calculated... | Download Scientific Diagram

ファイル:Properties of aqueous mixtures of pure salts. Thermodynamics of the ternary system- Water-sodium chloride-calcium chloride at 25 ?C (IA jresv70An4p313).pdf - Wikipedia

SOLVED: The observed vapor pressure lowering of a calcium chloride solution is 5.00 Torr at 20.0 degrees celsius. Calculate the molality of the calcium chloride in the solution.
![Vapor-Pressure Measurement of Ternary System CaCl2 + [Emim]Br + H2O, CaCl2 + [Emim]Cl + H2O, CaCl2 + [Emim]Ac + H2O, and CaCl2 + [Emim]NO3 + H2O,Journal of Chemical & Engineering Data - X-MOL Vapor-Pressure Measurement of Ternary System CaCl2 + [Emim]Br + H2O, CaCl2 + [Emim]Cl + H2O, CaCl2 + [Emim]Ac + H2O, and CaCl2 + [Emim]NO3 + H2O,Journal of Chemical & Engineering Data - X-MOL](https://xpic.x-mol.com/20210115%2F10.1021_acs.jced.0c00829.gif)
Vapor-Pressure Measurement of Ternary System CaCl2 + [Emim]Br + H2O, CaCl2 + [Emim]Cl + H2O, CaCl2 + [Emim]Ac + H2O, and CaCl2 + [Emim]NO3 + H2O,Journal of Chemical & Engineering Data - X-MOL

Vapor pressure of magnesium in the reduction of magnesium compounds... | Download Scientific Diagram
![Vapor-Pressure Measurement of Ternary System CaCl2 + [Emim]Br + H2O, CaCl2 + [Emim]Cl + H2O, CaCl2 + [Emim]Ac + H2O, and CaCl2 + [Emim]NO3 + H2O | Journal of Chemical & Engineering Data Vapor-Pressure Measurement of Ternary System CaCl2 + [Emim]Br + H2O, CaCl2 + [Emim]Cl + H2O, CaCl2 + [Emim]Ac + H2O, and CaCl2 + [Emim]NO3 + H2O | Journal of Chemical & Engineering Data](https://pubs.acs.org/cms/10.1021/acs.jced.0c00829/asset/images/medium/je0c00829_0008.gif)
Vapor-Pressure Measurement of Ternary System CaCl2 + [Emim]Br + H2O, CaCl2 + [Emim]Cl + H2O, CaCl2 + [Emim]Ac + H2O, and CaCl2 + [Emim]NO3 + H2O | Journal of Chemical & Engineering Data
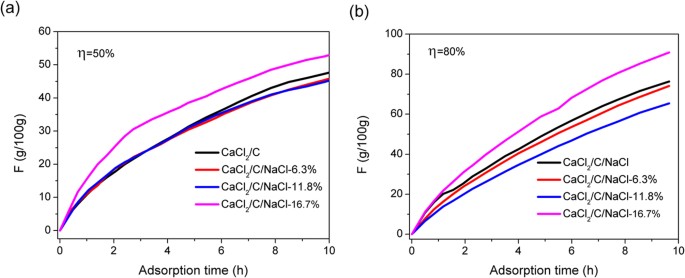

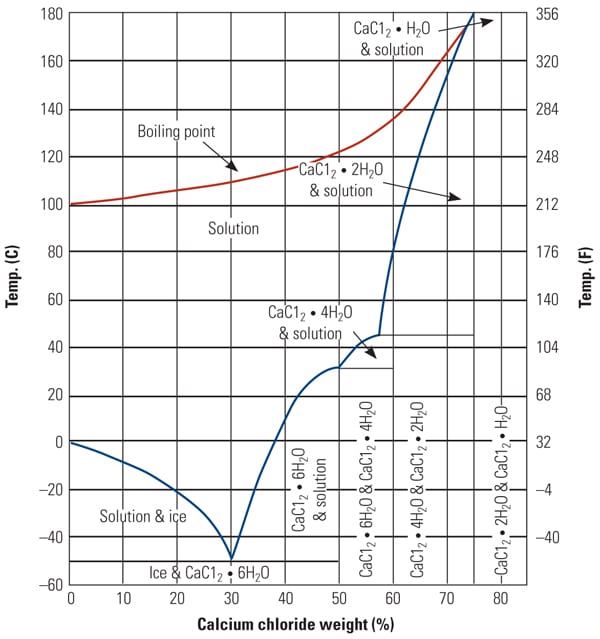
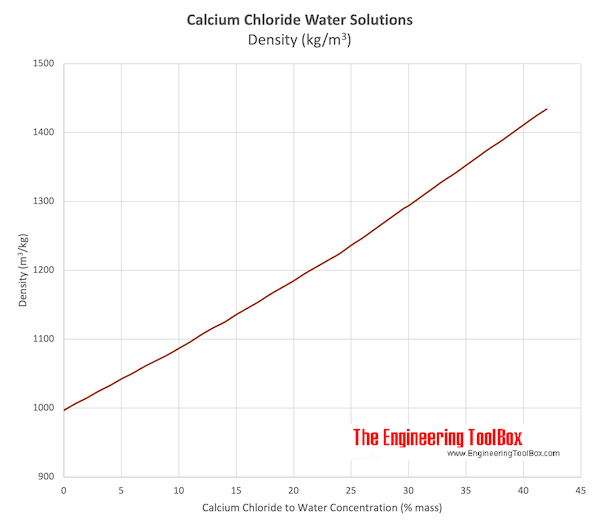

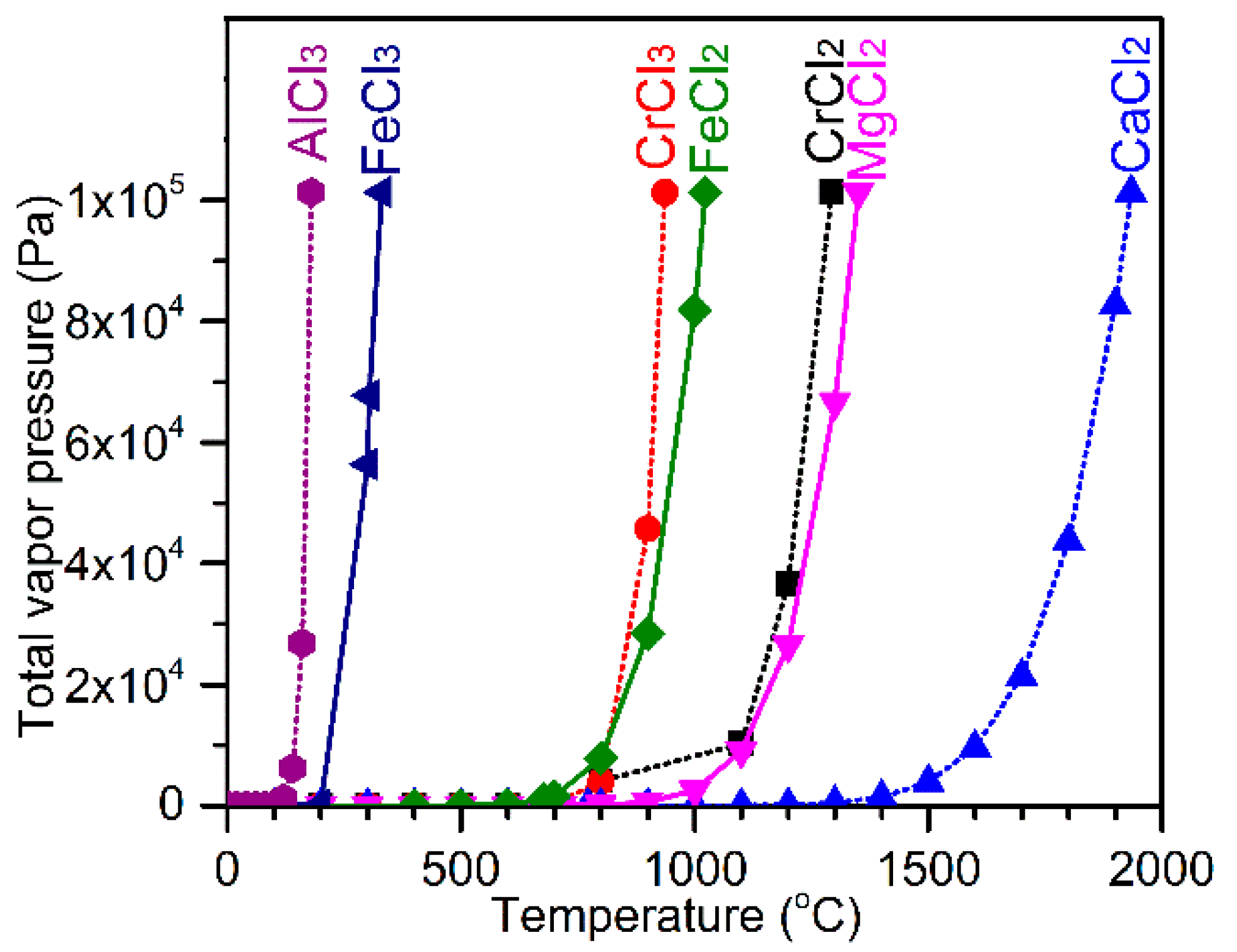
![Evolution of vapor pressure of several chlorides [32]. | Download Scientific Diagram Evolution of vapor pressure of several chlorides [32]. | Download Scientific Diagram](https://www.researchgate.net/publication/229383363/figure/fig2/AS:489936240549890@1493821310993/Evolution-of-vapor-pressure-of-several-chlorides-32.png)