VILNIAUS UNIVERSITETAS Rita Bukšnaitienė HETEROCIKLINIŲ JUNGINIŲ, GRETIMOSE PADĖTYSE TURINČIŲ ETINIL– IR FORMILFRAGMENT

Ruthenium-Catalyzed Cyclization of Epoxide with a Tethered Alkyne: Formation of Ketene Intermediates via Oxygen Transfer from Epoxides to Terminal Alkynes | Journal of the American Chemical Society

DNA fragmentation induced in HeLa cells by DMSO (control), Taxol (25... | Download Scientific Diagram

Biomolecules | Free Full-Text | Bioconversion of Biologically Active Indole Derivatives with Indole-3-Acetic Acid-Degrading Enzymes from Caballeronia glathei DSM50014 | HTML
![Study on the cyclization of 6-arylethynylpyrimidine-5-carbaldehydes with tert-butylamine: microwave versus thermal preparation of pyrido[4,3-d]pyrimidines - ScienceDirect Study on the cyclization of 6-arylethynylpyrimidine-5-carbaldehydes with tert-butylamine: microwave versus thermal preparation of pyrido[4,3-d]pyrimidines - ScienceDirect](https://ars.els-cdn.com/content/image/1-s2.0-S0040402009007145-fx1.jpg)
Study on the cyclization of 6-arylethynylpyrimidine-5-carbaldehydes with tert-butylamine: microwave versus thermal preparation of pyrido[4,3-d]pyrimidines - ScienceDirect

Study on the Reactions of Acetylenic Aldehydes with Dimethyl Phosphite in Basic Media: Phosphonate‐Phosphate Rearrangement versus 5‐exo‐dig Cyclization Reactions - Cikotiene - 2012 - Advanced Synthesis & Catalysis - Wiley Online Library

An Enolate‐Structure‐Enabled Anionic Cascade Cyclization Reaction: Easy Access to Complex Scaffolds with Contiguous Six‐, Five‐, and Four‐Membered Rings - Javorskis - 2020 - Angewandte Chemie International Edition - Wiley Online Library
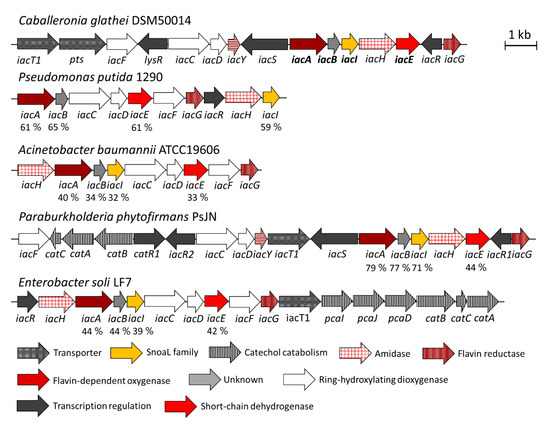
Biomolecules | Free Full-Text | Bioconversion of Biologically Active Indole Derivatives with Indole-3-Acetic Acid-Degrading Enzymes from Caballeronia glathei DSM50014 | HTML

An Enolate‐Structure‐Enabled Anionic Cascade Cyclization Reaction: Easy Access to Complex Scaffolds with Contiguous Six‐, Five‐, and Four‐Membered Rings - Javorskis - 2020 - Angewandte Chemie - Wiley Online Library
Bioconversion of Biologically Active Indole Derivatives with Indole-3-Acetic Acid-Degrading Enzymes from Caballeronia glathei DSM50014. - Document - Gale Academic OneFile

An Enolate‐Structure‐Enabled Anionic Cascade Cyclization Reaction: Easy Access to Complex Scaffolds with Contiguous Six‐, Five‐, and Four‐Membered Rings - Javorskis - 2020 - Angewandte Chemie - Wiley Online Library
![Lewis Acid-Catalyzed [4 + 2] Benzannulation between Enynal Units and Enols or Enol Ethers: Novel Synthetic Tools for Polysubstituted Aromatic Compounds Including Indole and Benzofuran Derivatives | The Journal of Organic Chemistry Lewis Acid-Catalyzed [4 + 2] Benzannulation between Enynal Units and Enols or Enol Ethers: Novel Synthetic Tools for Polysubstituted Aromatic Compounds Including Indole and Benzofuran Derivatives | The Journal of Organic Chemistry](https://pubs.acs.org/cms/10.1021/jo060597m/asset/images/large/jo060597mn00001.jpeg)
Lewis Acid-Catalyzed [4 + 2] Benzannulation between Enynal Units and Enols or Enol Ethers: Novel Synthetic Tools for Polysubstituted Aromatic Compounds Including Indole and Benzofuran Derivatives | The Journal of Organic Chemistry

Study on the Reactions of Acetylenic Aldehydes with Dimethyl Phosphite in Basic Media: Phosphonate‐Phosphate Rearrangement versus 5‐exo‐dig Cyclization Reactions - Cikotiene - 2012 - Advanced Synthesis & Catalysis - Wiley Online Library
Flow cytometric analysis of HeLa cells. a) Cell-cycle histograms of... | Download Scientific Diagram

Western blots obtained after exposing HeLa cells to Taxol (12 nM), 7.7... | Download Scientific Diagram

Electrophile‐Mediated Reactions of Functionalized Propargylic Substrates - Urbanaitė - 2015 - European Journal of Organic Chemistry - Wiley Online Library
![Gold-Catalyzed Cascade Annulations of 2-(Ynol)aryl Aldehydes: Facile Synthesis of Benzochromanes and Benzobicyclo[n.3.1]acetals | Organic Letters Gold-Catalyzed Cascade Annulations of 2-(Ynol)aryl Aldehydes: Facile Synthesis of Benzochromanes and Benzobicyclo[n.3.1]acetals | Organic Letters](https://pubs.acs.org/cms/10.1021/ol101985d/asset/images/medium/ol-2010-01985d_0001.gif)
Gold-Catalyzed Cascade Annulations of 2-(Ynol)aryl Aldehydes: Facile Synthesis of Benzochromanes and Benzobicyclo[n.3.1]acetals | Organic Letters
Tvirtinu....................................... VU MF Dekanas prof. A. Utkus 2021 m. gegužės mėn. d. VU MF farmacijos st




