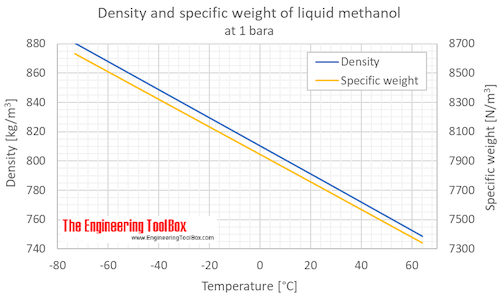
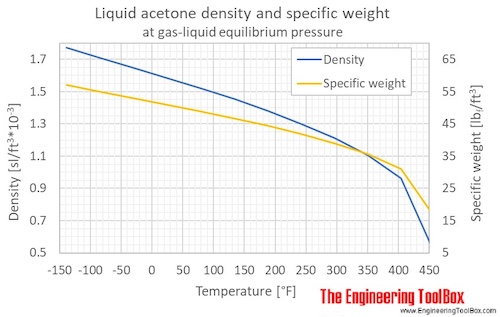
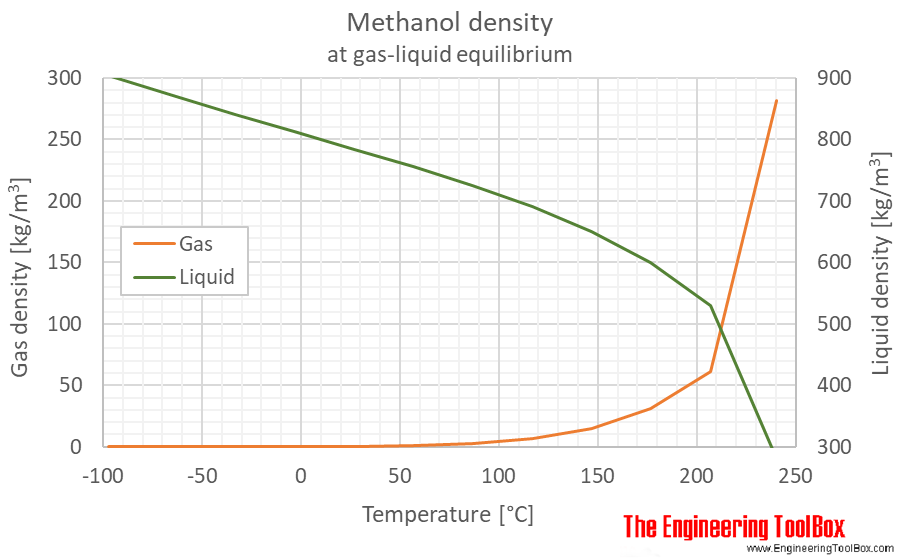
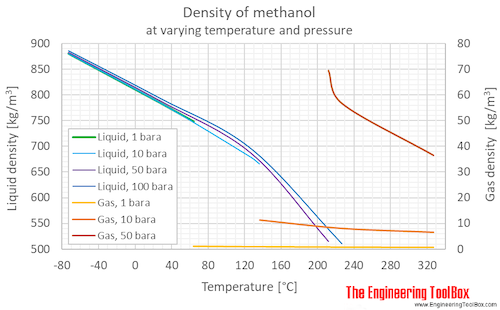
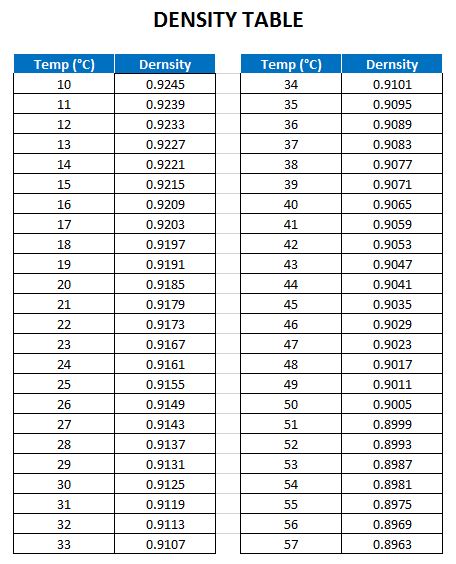
SOLVED:The density of acetonitrile (CH3 CN) is 0.786 g / mL and the density of methanol (CH3 OH) is 0.791 g / mL . A solution is made by dissolving 22.5 mL

Saturated-liquid and -vapor density differences, ρ S = ρ S − ρ S , and... | Download Scientific Diagram