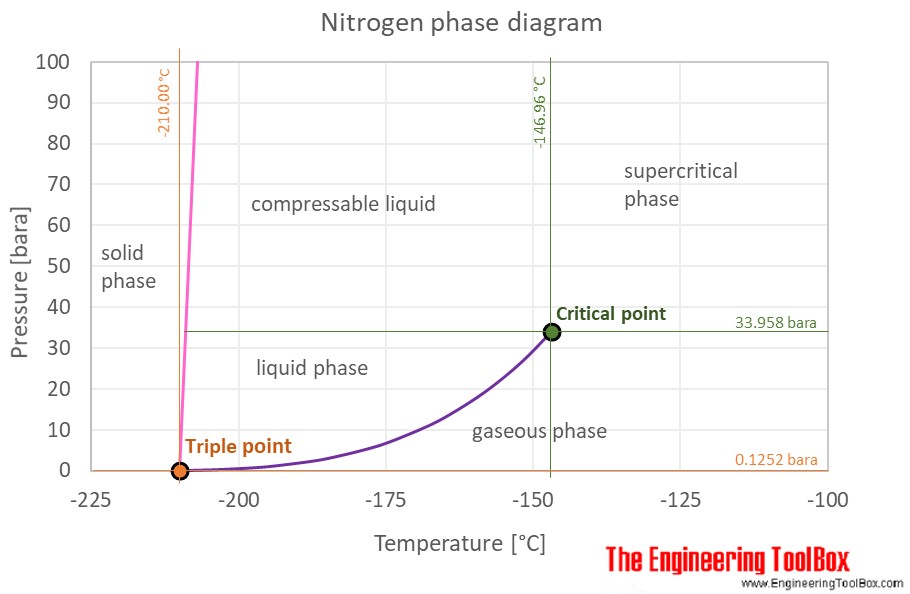
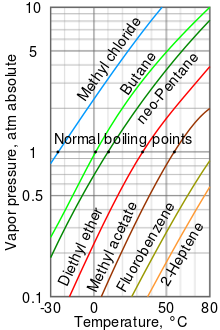
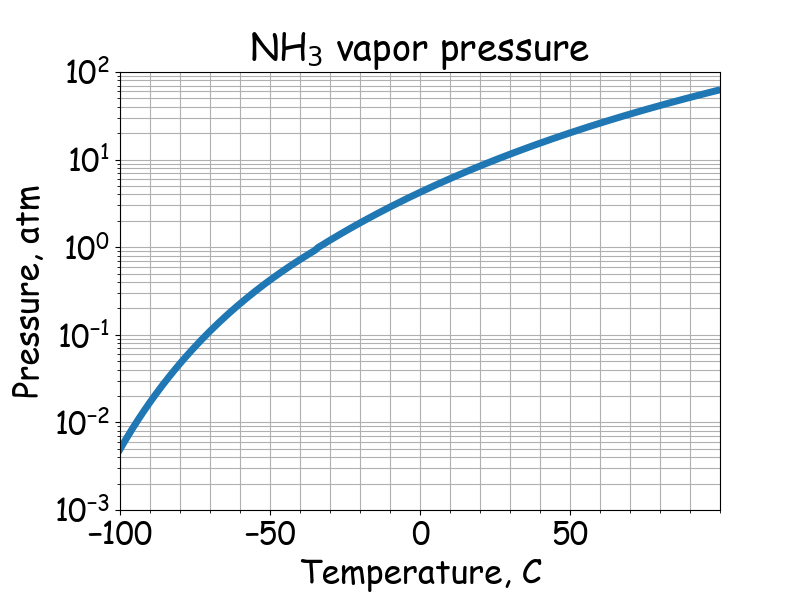
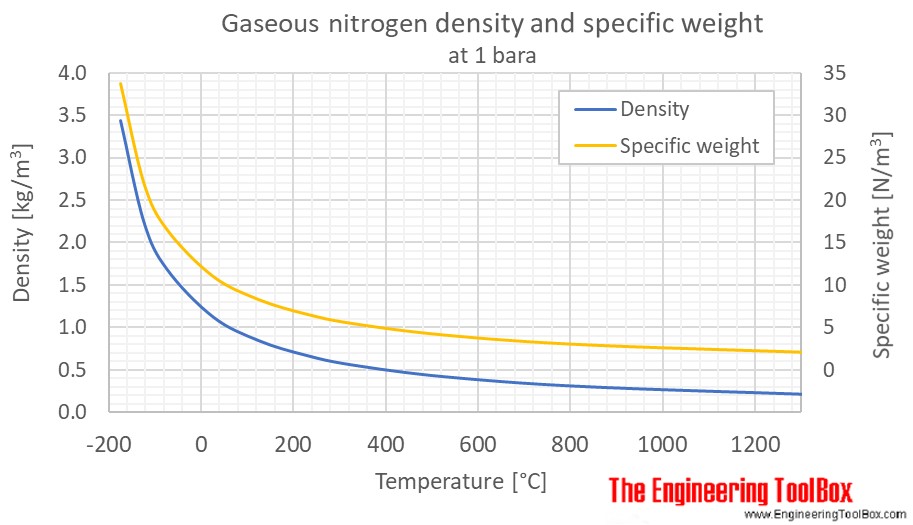
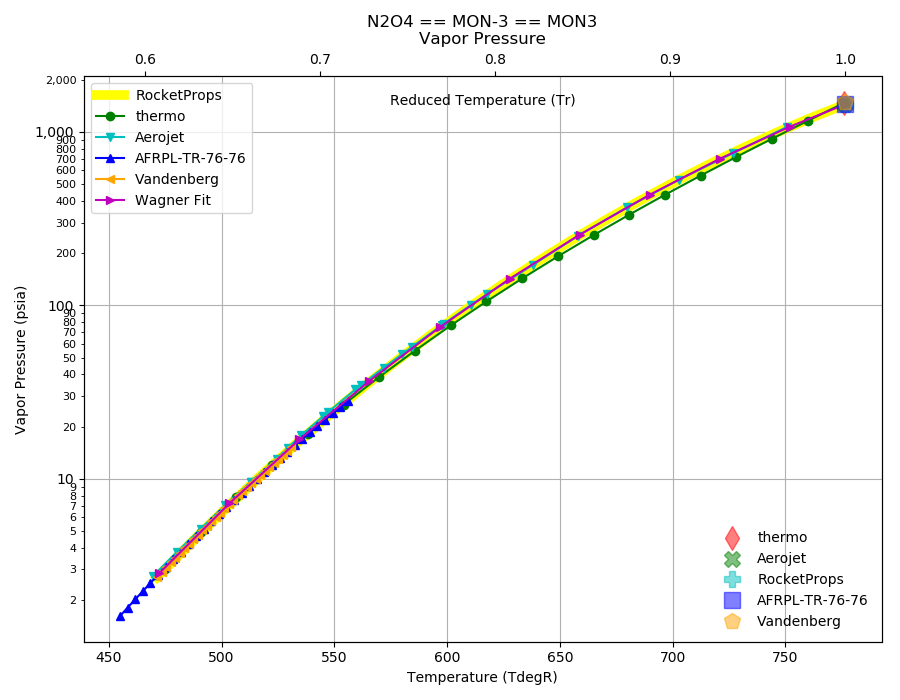
Liquid nitrogen has a boiling point of -195.81^0C atmospheric pressure. Express this temperature (a) in degrees Fahrenheit and (b) in kelvins.
Vapor pressures of concentrated nitric acid solutions in the composition range 83 to 97 percent nitric acid 0 to 6 percent nitrogen dioxide, 0 to 15 percent water, and in the temperature

Vapour pressure curves of nitrogen and air. 1 – air-evaporation curve,... | Download Scientific Diagram

Effect of nitrogen partial pressure on nitrogen solubility in liquid... | Download Scientific Diagram

Measurement and Correlation of the Vapor Pressure of a Series of α-Pinene Derivatives | Journal of Chemical & Engineering Data
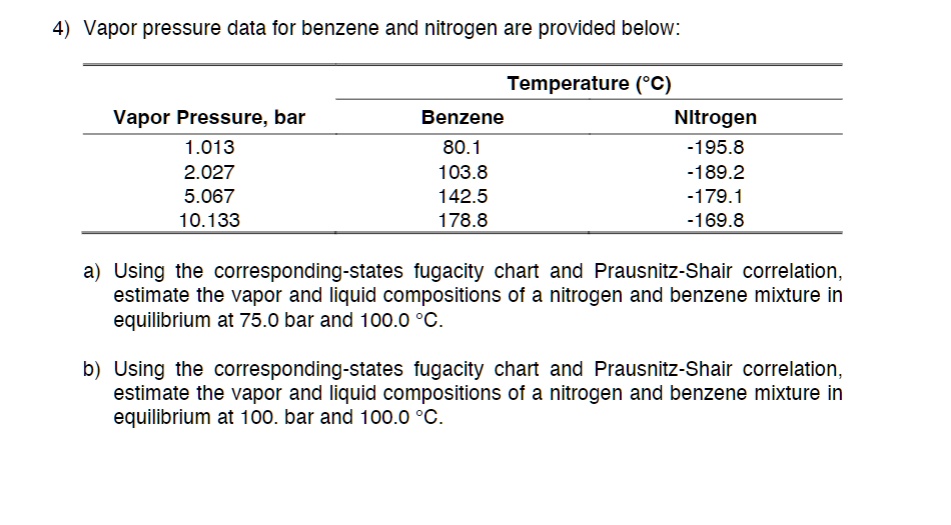
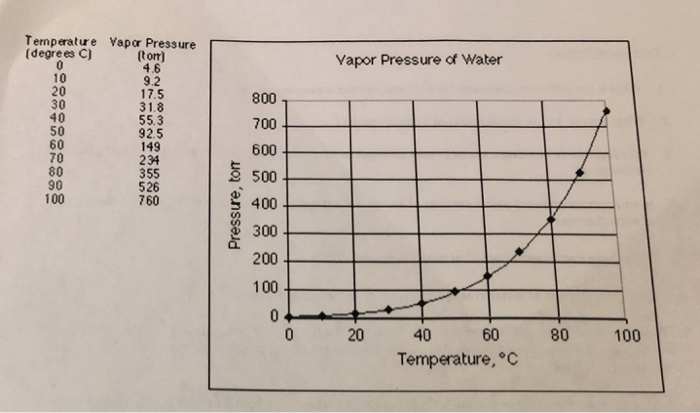
SOLVED: Texts: 4) Vapor pressure data for benzene and nitrogen are provided below: Temperature ('C) Vapor Pressure (bar): 1.013, 2.027, 5.067, 10.133 Benzene: 80.1, 103.8, 142.5, 178.8 Nitrogen: -195.8, -189.2, -179.1, -169.8

A sample of nitrogen is collected over water at 21 degrees Celsius at a pressure of 745 torr. What is the partial pressure, in torr, of the nitrogen? | Homework.Study.com
Vapor pressures and calculated heats of vaporization of concentrated nitric acid solutions in the composition range 71 to 89 percent nitrogen dioxide, 1 to 10 percent water, and in the temperature range
Vapor pressures of concentrated nitric acid solutions in the composition range 83 to 97 percent nitric acid 0 to 6 percent nitrogen dioxide, 0 to 15 percent water, and in the temperature